The Apple iPad 2 Review
by Brian Klug, Anand Lal Shimpi & Vivek Gowri on March 19, 2011 8:01 PM ESTI asked a friend of mine, Alexander Miles, to write a bit about the properties of glass that really contribute to its overall strength after reading that the iPad 2's glass is 0.62 mm thick compared to 0.85 mm thick in the iPad 1. Hopefully this dispels some myths about glass strength and clarifies. Alex is a senior double majoring in Materials Science and Engineering and Optical Science and Engineering at the University of Arizona.
On the Strength of Glass
We usually think of things failing under compressive stress, being pushed inward from both sides until it they are crushed. Glass and ceramics, it turns out, are incredibly strong in compressive stress. Strictly by the numbers, a fire truck could be supported by a ceramic coffee cup underneath each tire, but only if the load was perfectly downward. Why then are glasses so fragile? This is because no situation causes only compressive stresses, and tensile stress (imagine pulling something from both ends) is what causes glasses to fail. One can think of tiny cracks inside the glass being pushed closed under compressive stress, but torn open under tensile stress.
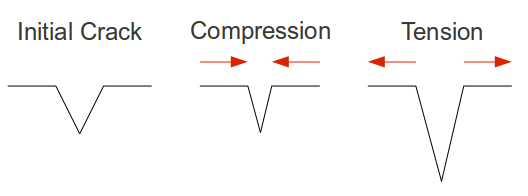
A schematic illustration of crack behavior in brittle materials.
If you test the tensile strength of thousands of pieces of glass with identical processing and geometry, you will get thousands of different answers. This is markedly different from metals, where you will get nearly the same result every time. The reason being that glass and ceramic materials have a much lower fracture toughness, as much as 100 times smaller than that of a metal. Fracture toughness indicates how easily a crack can propagate, or to phrase it differently, how big a flaw will cause fracture for a given load. As the required load for normal flaw sizes in metals is enormous, metals typically do not fracture in the way glasses do. Metals usually fail in what is called plastic deformation, necking down then tearing away, long before fracture can occur. This plastic deformation is very predictable and follows the stress-strain curve for the given metal, whereas glasses are less predictable.
The question now is, how does the size of a piece of glass affect its behavior under tensile stress? It depends on the distribution of flaws within the material. If you strike a piece of glass with a hammer, a compressive stress is created right below the hammer, but a ring of tensile stress is also created around the spot you hit. You are essentially sampling the distribution of flaws, because if any of the flaws in the affected glass are big enough to widen with the stress you provided, they will rapidly propagate and the material will fracture. The stress field extends down into he material, so flaws in the volume can cause failure as well, though surface flaws are more consistently to blame as the stresses encountered there are almost always larger.
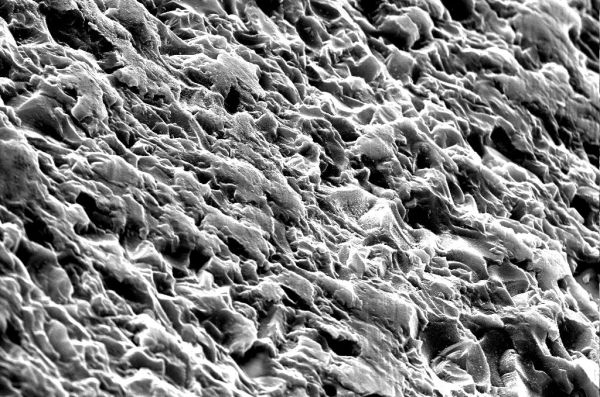
SEM image of a broken glass surface, (CC BY-NC-ND 2.0) attribution St Stev's flickr.
In glasses the distribution of failure stresses is described using Weibull statistics, giving a peak where most samples fail, and tails both on the high and low end where samples had abnormally high and low failure stresses respectively. The long and short of this is that consistently processed samples will have a more narrow distribution as the geometries are scaled down. This means that a thinner piece of glass will have fewer flaws in the bulk and far less likelihood of having a large enough flaw to cause catastrophic failure compared to a thicker piece of glass. The fact that the screens have a large aspect ratio, that is they are far thinner than they are wide or long, means that the effective stiffness will be different in the two directions. Taking this to an extreme, a very thin glass fiber is fairly flexible in bending, but very stiff axially, as its cross-section is so small that very few flaws are contained inside it. If one needs to break a glass fiber, a surface flaw is usually created by scratching it first.
| Approximate values for the strength of common soda lime glass in various conditions | |||||
| Condition | Tensile Strength (MPa) | ||||
| Theoretical Maximum (Flawless) | 9810 | ||||
| 3 Micron Fibers | 3330 | ||||
| Thin rods, fire-polished and acid etched | 3420 | ||||
| Thin rods, no special treatment | 690 | ||||
| Bulk, ion exchange tempered | 350 | ||||
| Bulk, thermally tempered | 300 | ||||
| Bulk, fire-polished and acid etched | 220 | ||||
| Bulk, no special treatment | 50 | ||||
In order to use glass screens on our devices, we would like it to be far tougher, where "tougher'' ideally means both more resistant to fracture as well as more resistant to scratching. There are two basic schemes used to strengthen glass: elimination of the surface flaws, and creation of compressive stress in the bulk of the glass. Eliminating the surface flaws by polishing, fire-polishing (heating them until surface tension flattens out the flaws), and acid-etching does indeed increase the strength, and drastically so. An increase in strength of up to one hundred times can result from such treatment, but is temporary as microscopic scratches from handling will quickly reduce the strength back to what it was before.
The second scheme for improving strength, introducing a compressive layer, works because existing compressive stress in the glass has to be overcome by the induced tensile stress before any cracks can propagate. To say it plainly, if you don't hit it hard enough with a hammer, it will not even see the type of stress that makes it fail. The down side to this method is that every force causes an equal and opposite force, meaning that a lot of compression at the surface causes tension at the center. As long as a crack does not reach the volume with the additional tensile stress imposed on it, the glass will hold together, but once it does it releases the energy kinetically and fails catastrophically (it explodes like a pumpkin with an M80 stuffed in it).
The way this layer is created varies based on the application. For car windshields, they are thermally tempered by chilling the outer surface while the center is still hot, as the surface remains solid while the center is still busy shrinking, which leaves the surface in compression. Similarly, coating the glass object in a second type of glass with a lower thermal expansion will cause the same effect, as the outer surface shrinks the center is shrinking faster. The multiple-glass approach has the additional benefit that cracks have difficultly moving from one type of glass to the next, leading Corning to produce some glasses with as many as 7 layers.
The final method, and most relevant to our discussion, is ion-exchange. Ion-exchange refers to removing small ions, like sodium, from the glass, and replacing them with larger ions like potassium, all at a temperature that prevents the structure of the glass from adjusting itself to these new bigger ions. The way this swap is actually done is by immersing the glass in a molten salt solution containing the ion we want to substitute in, and allowing it to diffuse in over time, while the smaller, more mobile, ion diffuses out. Depending upon the type of glass, the ions being exchanged, and the desired depth, this process can take as long as several days.
The iPad 2 and previous iPad both utilize Corning Gorilla Glass. This type of glass is an alkali-aluminosilicate, being primarily silica and aluminum with an alkali metal, along with other unspecified components mixed in to tweak its properties. The biggest benefit of alluminosilicate glasses, aside from being relatively tough to start with, is the fact that the rate of ion exchange is fairly high even at temperatures low enough that the structure cannot react, meaning it can be processed quickly and create deep protective layers in the glass. The iPad 2 has a modest reduction in the thickness of the glass (about 23% thinner, for those interested) compared to the first iPad, and the question of increased fracture risk has been posed. Given the identical surface quality between the two generations, the reduction of thickness should create no palpable change in toughness for the typical user. That is to say, a drop that would shatter the screen on the original iPad would likely do the same for the new model. That being said, several other design changes appear to account for the change, and might yield better performance in this department.
Where its predecessor used small metal clips to retain the glass screen, the current iteration uses a ring of adhesive around the entire perimeter that not only distributes the load around the glass and prevents scoring at the glass-metal interface, but better couples the stresses into the more compliant aluminum frame. Both of these measures should improve the performance; either way, drop-testing new electronics is generally not recommended.











189 Comments
View All Comments
claytontullos - Saturday, March 19, 2011 - link
http://technabob.com/blog/2011/03/18/ipad-2-refrig... kind of fun?vol7ron - Saturday, March 19, 2011 - link
This just goes back to what I've said since the iPad was introduced. It'll be the +1 device that's best for laying around your house. This goes into my review as why it needs to hit the $200-250 price point.Sure it's a nice e-Reader and can entertain with some games and even allow for some production work, but it is still clunky and uncomfortable and to be efficient and productive you need the additional hardware, which are going to bring you in a nice laptop range anyhow.
The 3GS is hitting the $50-100 price point w/ a 2 year contract, which I suggested a year ago. Personally, I still think that should be the price w/o the contract (to be available after-market for gifts/presents), but as long it's available at that point, that's where it needs to be.
I still think the iPad needs to drop to that $200-250 point. It's the coffee table device, which people should consider having 2-3 spread-out in the home [ maybe one in the bathroom ;) ] - if only they could also self-sync wirelessly. I'm not too sure who buys the base model, but the specs alone would keep me from considering it and when you look at the higher spec'd models, it's not as justified when looking at laptops, or other eReaders.
solipsism - Sunday, March 20, 2011 - link
$200 to $250 for a newly released 10” Tablet with an IPS panel? WTF are you smoking? How can you have such an odd mental disconnect between writing that and then writing "The 3GS is hitting the $50-100 price point w/ a 2 year contract”? What part of 2-year contract aren’t you understanding? Do you not realize the carrier is paying Apple more than $200-250 for that 3GS, and you are paying the carrier a lot more than that over 2 years?Pray tell, how would this device be $200-250 when the competition with a 2 decade head start still hasn’t been able to compete on price?
synaesthetic - Sunday, March 20, 2011 - link
I don't get how they sell so many when they're so useless and clunky... and cost so much.Lot of hipsters I guess.
michael2k - Sunday, March 20, 2011 - link
The weight, battery life, and cost (altogether) are unparalleled in the computing world.Smartphones with similar performance characteristics have far smaller screens and lower battery life.
PCs with similar battery life cost far more and weigh far more.
PCs with similar weight (and still double at that) cost far more and have only fraction of the battery life.
PCs with similar cost weigh far more and have drastically lower battery life.
Meaker10 - Sunday, March 20, 2011 - link
A dual core sandy bridge 13" device is going to be far more useful for work and far more powerful.michael2k - Sunday, March 20, 2011 - link
Who said anything about work? For things like reading Anandtech it would be far heavier, bulkier, and with less battery life.bigboxes - Sunday, March 20, 2011 - link
Just admit that it's a toy. The authors laid it out for you on how they prefer to use other devices instead of the iPad. It too bulky for portability and underpowered for any productivity tasks.So, you're telling me (and everyone else here) that you paid $500+ just to surf AnandTech on your couch? Just wondering.
Stas - Monday, March 21, 2011 - link
That's exactly why it cannot cost this much to be a reasonable buy. No, the following purchases are not reasonable: fa- sheep base, soccer moms that buy the latest gadget with most hype for their kids/husbands not even knowing wtf it does, or PR boost in form of including, again, the most hyped device with cars, hotel rooms, air travel, etc (3 categories right there probably account for 90% of all sales). I mean people that understand exactly what the device is, what it's not, and have a clear idea of how they are going to use it. And it doesn't matter how much it costs to make it, how advanced the hardware is, or how "revolutionary" the design is. Given the limited usability of a slim, touchscreen device, I think asking $600+ for one is ballsy.MScrip - Monday, March 21, 2011 - link
-- "Given the limited usability of a slim, touchscreen device, I think asking $600+ for one is ballsy." --That's true about any tablet.
As great as Honeycomb tablets are... they're still not gonna provide a true computing experience.
A $600 laptop will always provide far more functionality than a $600 tablet...
Yet... all these manufacturers are pumping out tablets at an alarming rate.
Apple took the risk and added a new product to their lineup.
If tablets were destined to fail... we wouldn't see Motorola, Samsung and even RIM jumping into the tablet game...